Talk by Dr Clara Barker Monday 20 July 2020
https://twitter.com/ClaraMBarker
https://en.wikipedia.org/wiki/Clara_Barker
http://users.ox.ac.uk/~engs1643/member_clara_barker.html
https://www.huffingtonpost.co.uk/entry/lgbt-science_uk_5a8ef065e4b0746ba2acf7e5?guccounter=1
In this talk, Dr Barker provided an introduction to superconductors, their properties and main uses. She also discussed how her group produce thin film superconductors in the Centre for Applied Superconductivity as Oxford University, using pulsed laser deposition and magnetron sputtering. She also touched on her advocacy as a scientist who is also transgender, and why we must strive for active inclusivity in STEM fields.
The following are notes from the on-line lecture. Even though I could stop the video and go back over things there are likely to be mistakes because I haven’t heard things correctly or not understood them. I hope Dr Barker and my readers will forgive any mistakes and let me know what I got wrong.
What is material science?
It is a subject that involves physics (hooray), engineering and chemistry (Boo)
Let’s use carbon to illustrate the subject.
Carbon has several allotropes
https://en.wikipedia.org/wiki/Allotropy
Allotropy or allotropism (from Ancient Greek ἄλλος (allos), meaning ‘other’, and τρόπος (tropos), meaning ‘manner, form’) is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Allotropes are different structural modifications of an element; the atoms of the element are bonded together in a different manner.
The term allotropy is used for elements only, not for compounds. The more general term, used for any crystalline material, is polymorphism. Allotropy refers only to different forms of an element within the same phase (i.e.: solid, liquid or gas states); the differences between these states would not alone constitute examples of allotropy.
For some elements, allotropes have different molecular formulae despite difference in phase; for example, two allotropes of oxygen (dioxygen, O2, and ozone, O3) can both exist in the solid, liquid and gaseous states. Other elements do not maintain distinct allotropes in different phases; for example, phosphorus has numerous solid allotropes, which all revert to the same P4 form when melted to the liquid state.
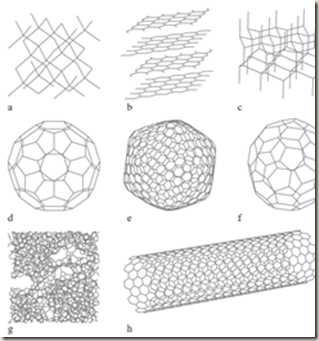
The allotropes of carbon include diamond (the carbon atoms are bonded together in a tetrahedral lattice arrangement), graphite (the carbon atoms are bonded together in sheets of a hexagonal lattice), graphene (single sheets of graphite), and fullerenes (the carbon atoms are bonded together in spherical, tubular, or ellipsoidal formations).
https://en.wikipedia.org/wiki/Allotropes_of_carbon
Carbon is capable of forming many allotropes (structurally different forms of the same element) due to its valency. Well-known forms of carbon include diamond and graphite.
In recent decades, many more allotropes have been discovered and researched including ball shapes such as buckminsterfullerene and sheets such as graphene. Larger scale structures of carbon include nanotubes, nanobuds and nanoribbons. Other unusual forms of carbon exist at very high temperatures or extreme pressures. Around 500 hypothetical 3-periodic allotropes of carbon are known at the present time, according to the Samara Carbon Allotrope Database (SACADA).
Eight allotropes of carbon: a) diamond, b) graphite, c) lonsdaleite, d) C60 buckminsterfullerene, e) C540, Fullerite f) C70, g) amorphous carbon, h) zig-zag single-walled carbon nanotube. Missing: cyclocarbon, carbon nanobuds and schwarzites.
Material science involves making changes to the structure of a substance on the nano (small) scale and/or macro (big) scale
Superconductors: Electrical properties
Metals are, generally, excellent conductors because of their structure.
They consist of a metallic lattice
Each atom will have 1 or 2 electrons in their outer shell which can become delocalised and free
Applying a potential difference across the metal makes these electrons move through the conductor and an electric current is formed
Charge carriers flowing = current.
Now ordinarily these electrons don’t have an easy route through the metal. The atoms (technically positive ions) vibrate and provide obstacles to the electrons.
The electrons collide with the atoms transferring energy (the resistance to current flow) and this gives us useful things such as heat (electric cooker) and light and non-useful things such as heat (overheating electric motor)
Cooling the conductor down reduces the resistance. Atoms vibrate less and less.
Cooling the conductor down below a critical temperature reduces the resistance completely.
Below the critical temperature, Tc, electrons team up in pairs in some superconductors. These pairs are called cooper pairs
http://www.supraconductivite.fr/en/index.php?p=supra-explication-cooper-more
How can electrons form pairs while they should be repelling each other? Indeed, electrons are electrically negative, and two negative charges repel each other. Cooper gives the following explanation:
http://www.supraconductivite.fr/media/videos/cooper-basseT.webm
At a low temperature, when an electron moves, it slightly attracts the surrounding atoms. The reason is that the electron has a negative electric charge and the atoms a positive one, since the latter is missing one of its electrons (we should say “positive ions” rather than “atoms”). A negative charge attracts a positive charge. If a second electron appears at that moment, it experiences two atoms at a closer range than usual for a short period of time, and it will be pulled between them. Two atoms closer together create a more electrically positive zone, thus being more attractive to the electron. The second electron naturally follows the first due to the oscillation of atoms. This is called “attraction via network vibrations” (or “phonons”).
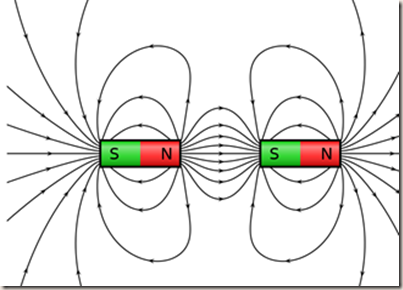
We can use bar magnets to help explain what is going on.
Magnetic field lines run from north to south. When you bring magnets together these magnetic fields interact. North and south poles attract each other and the magnets pull together.
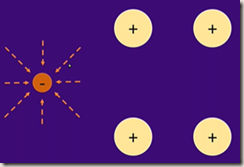
Electric charges produce a similar pattern. Electric field lines run from positive to negative,
Electron electric field interacts with the positive ion electric field
The positive area around the charge pulls the electron in and the second electron is pulled in.
https://en.wikipedia.org/wiki/BCS_theory
BCS theory or Bardeen–Cooper–Schrieffer theory (named after John Bardeen, Leon Cooper, and John Robert Schrieffer) is the first microscopic theory of superconductivity since Heike Kamerlingh Onnes’s 1911 discovery. The theory describes superconductivity as a microscopic effect caused by a condensation of Cooper pairs. The theory is also used in nuclear physics to describe the pairing interaction between nucleons in an atomic nucleus.
It was proposed by Bardeen, Cooper, and Schrieffer in 1957; they received the Nobel Prize in Physics for this theory in 1972.
https://en.wikipedia.org/wiki/John_Bardeen (below left)
John Bardeen (May 23, 1908 – January 30, 1991) was an American physicist.
https://en.wikipedia.org/wiki/Leon_Cooper (Above centre)
Leon N. Cooper (born February 28, 1930) is an American physicist and Nobel Prize laureate who, with John Bardeen and John Robert Schrieffer, developed the BCS theory of superconductivity. He is also the namesake of the Cooper pair and co-developer of the BCM theory of synaptic plasticity.
https://en.wikipedia.org/wiki/John_Robert_Schrieffer (Above right)
John Robert Schrieffer (May 31, 1931 – July 27, 2019) was an American physicist who, with John Bardeen and Leon Cooper, was a recipient of the 1972 Nobel Prize in Physics for developing the BCS theory, the first successful quantum theory of superconductivity.
https://en.wikipedia.org/wiki/Heike_Kamerlingh_Onnes
Professor Heike Kamerlingh Onnes, (21 September 1853 – 21 February 1926) was a Dutch physicist and Nobel laureate. He discovered superconductivity in 1911.
https://en.wikipedia.org/wiki/Superconductivity
Superconductivity is the set of physical properties observed in certain materials where electrical resistance vanishes and magnetic flux fields are expelled from the material. Any material exhibiting these properties is a superconductor. Unlike an ordinary metallic conductor, whose resistance decreases gradually as its temperature is lowered even down to near absolute zero, a superconductor has a characteristic critical temperature below which the resistance drops abruptly to zero. An electric current through a loop of superconducting wire can persist indefinitely with no power source.
Apply a potential difference to a superconductor in a closed loop and the current produced will continue to flow when you remove the potential difference, as long as it remains cold. Cold persistent current
https://en.wikipedia.org/wiki/Electron_magnetic_resonance
In physics, biology and chemistry, electron magnetic resonance (EMR) is an interdisciplinary field with several forms: electron paramagnetic resonance (EPR), electron spin resonance (ESR) and electron cyclotron resonance (ECR). EMR looks at electrons rather than nuclei or ions as in nuclear magnetic resonance (NMR) and ion cyclotron resonance (ICR) respectively.
EMR is used to investigate superconductors.
Now a major problem is that too much current will cause copper (the usual material for electrical wiring) to melt. To get over this “problem” increase the cross-sectional area of the wiring. However, this causes another problem. Copper cables take up a lot of space.
With superconductors there is zero resistance. The current is high but the heating effect isn’t present so the copper wires can be much thinner.
https://energytransition.org/2014/11/worlds-longest-superconductor-in-operation-in-germany/
https://www.en-former.com/en/grid-expansion-series-superconductor-cables/
Superconductors are part of everyday life in Essen.
https://en.wikipedia.org/wiki/Essen
https://ec.europa.eu/energy/sites/ener/files/documents/18._eoin_-_supernode_horizon_2050.pdf
http://gsr.to.infn.it/wp-content/uploads/sites/8/2017/06/Cogne-Part-I_Flukiger.pdf
In order to reach the temperature of -200 degrees Celsius, the cables are sheathed in an insulating sleeve that carries liquid nitrogen. The big problem with this system is that the cables have to be kept cold. This is expensive.
There is also an upper limit to the size of the current that can flow through the superconductor. Critical current density Jc is the maximum current a superconducting wire can carry per unit cross-sectional area
https://en.wikipedia.org/wiki/Current_density
Superconductors have magnetic properties and magnet can levitate above a bed of magnets
https://www.unf.edu/~michael.lufaso/chem4627/ch9_solid_state.pdf
Type I Superconductors:
Superconductivity exists only below a critical temperature and below a critical magnetic field strength. • “Soft superconductors”
The Meissner effect (or Meissner–Ochsenfeld effect) is the expulsion of a magnetic field from a superconductor during its transition to the superconducting state when it is cooled below the critical temperature.
Type II Superconductors
Type II ‘hard’ superconductors have much higher critical fields and carry much higher current densities while remaining in the superconducting state.
In type-I superconductors, superconductivity is abruptly destroyed when the strength of the applied field rises above a critical value Hc. Depending on the geometry of the sample, one may obtain an intermediate state consisting of a baroque pattern of regions of normal material carrying a magnetic field mixed with regions of superconducting material containing no field.
In type-II superconductors, raising the applied field past a critical value Hc1 leads to a mixed state (also known as the vortex state) in which an increasing amount of magnetic flux penetrates the material, but there remains no resistance to the electric current as long as the current is not too large. At a second critical field strength Hc2, superconductivity is destroyed. The mixed state is caused by vortices in the electronic superfluid, sometimes called fluxons because the flux carried by these vortices is quantized. Most pure elemental superconductors, except niobium and carbon nanotubes, are type I, while almost all impure and compound superconductors are type II.
Type-II superconductors are more complicated than type-I
As it cools down the quantised field lines pass through particular areas of the superconductor. Very good at trapping fields.
Moving the bar magnet makes the superconductor stay in the same relative position.
The image below shows superconductor in a cup of liquid nitrogen. Moving the superconductor moves the magnet as they want to stay in the same relative position
The image above shows a superconducting puck floating above a bed of magnets. You can turn the whole thing upside down and the puck will be suspended rather than levitating.
https://www.youtube.com/watch?v=zPqEEZa2Gis
History, material and uses
As mentioned earlier superconductivity was discovered in 1911 by Professor Heike Kamerlingh Onnes.
https://en.wikipedia.org/wiki/History_of_superconductivity
Onnes began the investigations with platinum and gold, replacing these later with mercury (a more readily refinable material). Onnes’s research into the resistivity of solid mercury at cryogenic temperatures was accomplished by using liquid helium as a refrigerant. On April 8, 1911, 16:00 hours Onnes noted “Kwik nagenoeg nul”, which translates as “[Resistance of] mercury almost zero.” At the temperature of 4.19 K, he observed that the resistivity abruptly disappeared (the measuring device Onnes was using did not indicate any resistance). Onnes disclosed his research in 1911, in a paper titled “On the Sudden Rate at Which the Resistance of Mercury Disappears.” Onnes stated in that paper that the “specific resistance” became thousands of times less in amount relative to the best conductor at ordinary temperature. Onnes later reversed the process and found that at 4.2 K, the resistance returned to the material. The next year, Onnes published more articles about the phenomenon. Initially, Onnes called the phenomenon “supraconductivity” (1913) and, only later, adopted the term “superconductivity.” For his research, he was awarded the Nobel Prize in Physics in 1913.
Onnes conducted an experiment, in 1912, on the usability of superconductivity. Onnes introduced an electric current into a superconductive ring and removed the battery that generated it. Upon measuring the electric current, Onnes found that its intensity did not diminish with the time. The current persisted due to the superconductive state of the conductive medium.
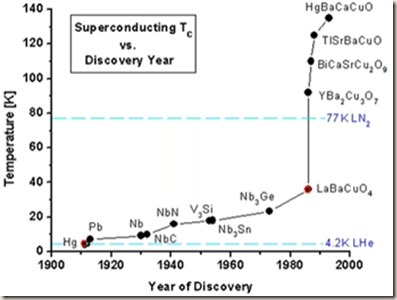
In subsequent decades, superconductivity was found in several other materials; In 1913, lead at 7 K, in 1930’s niobium at 10 K, and in 1941 niobium nitride at 16 K.
In 1985 certain ceramics were turned from insulators to superconductors at 90K. The advantage here is that liquid nitrogen could be used for cooling instead of liquid helium. Liquid nitrogen is much more available.
https://en.wikipedia.org/wiki/Magnesium_diboride
Magnesium diboride is the inorganic compound with the formula MgB2. It is a dark grey, water-insoluble solid. The compound has attracted attention because it becomes superconducting at 39 K (−234 °C). In terms of its composition, MgB2 differs strikingly from most low-temperature superconductors, which feature mainly transition metals. Magnesium diboride was actually considered a waste product.
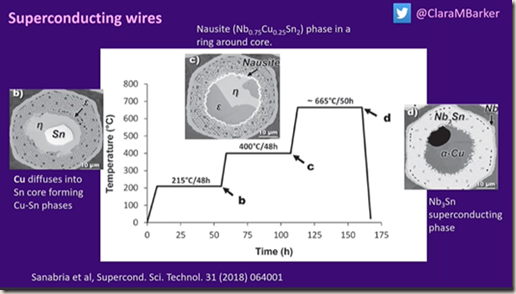
Manufacture
Wrapping in Nb foil to stop any reaction with copper. Extrude into wires a few mm across.
Process results in a 1.5mm wide 36 strands. Each strand is made up of 3600 smaller filaments 6mm in diameter. You need to use an electron microscope to see the arrangement.
Niobium tin will be used in ITER and the CERN upgrade. The arrangement is brittle so you extrude with tin before introducing the niobium.
Treating with heat changes the phase and properties.
Not all phases of Nb2Sn is superconducting
https://en.wikipedia.org/wiki/ITER
ITER (originally the International Thermonuclear Experimental Reactor) is an international nuclear fusion research and engineering megaproject, which will be the world’s largest magnetic confinement plasma physics experiment. It is an experimental tokamak nuclear fusion reactor that is being built next to the Cadarache facility in Saint-Paul-lès-Durance, in Provence, southern France.
https://home.cern/science/accelerators/large-hadron-collider
The Large Hadron Collider (LHC) is the world’s largest and most powerful particle acceleratorThe LHC consists of a 27-kilometre ring of superconducting magnets with a number of accelerating structures to boost the energy of the particles along the way.
The European Organisation for Nuclear Research (CERN) is in the middle of giving the Large Hadron Collider (LHC), the most powerful particle accelerator in the world, a major upgrade.
The HL-LHC upgrade is designed to increase the collider’s luminosity increase by a factor of 10 over the original machine. This requires a significant modification to the beam line around the two largest LHC detectors – ATLAS and CMS. The work will involve upgrading about 1.2 km of the 27 km ring by including 11-12 T superconducting magnets and superconducting “crab” cavities – that reduce the angle at which the bunches cross – to increase the number of collisions at the two detectors. The upgrade also involves modifications to the LHC’s detector so that it can handle the increased luminosity.
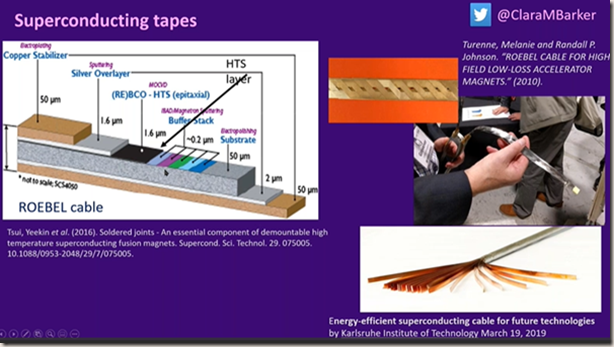
Make cables flexible by placing the parts in layers. Use 1% thickness superconductor
In Oxford they use a different system
Pulsed layer deposition system is used.
What is magnetron sputtering?
http://www.semicore.com/what-is-sputtering
https://en.wikipedia.org/wiki/Sputter_deposition
Sputter deposition is a physical vapour deposition (PVD) method of thin film deposition by sputtering. This involves ejecting material from a “target” that is a source onto a “substrate” such as a silicon wafer. Resputtering is re-emission of the deposited material during the deposition process by ion or atom bombardment. Sputtered atoms ejected from the target have a wide energy distribution, typically up to tens of eV (100,000 K). The sputtered ions (typically only a small fraction of the ejected particles are ionized — on the order of 1 percent) can ballistically fly from the target in straight lines and impact energetically on the substrates or vacuum chamber (causing resputtering). Alternatively, at higher gas pressures, the ions collide with the gas atoms that act as a moderator and move diffusively, reaching the substrates or vacuum chamber wall and condensing after undergoing a random walk. The entire range from high-energy ballistic impact to low-energy thermalized motion is accessible by changing the background gas pressure. The sputtering gas is often an inert gas such as argon. For efficient momentum transfer, the atomic weight of the sputtering gas should be close to the atomic weight of the target, so for sputtering light elements neon is preferable, while for heavy elements krypton or xenon are used. Reactive gases can also be used to sputter compounds. The compound can be formed on the target surface, in-flight or on the substrate depending on the process parameters. The availability of many parameters that control sputter deposition make it a complex process, but also allow experts a large degree of control over the growth and microstructure of the film.
https://en.wikipedia.org/wiki/High-power_impulse_magnetron_sputtering
High-power impulse magnetron sputtering (HIPIMS or HiPIMS, also known as high-power pulsed magnetron sputtering, HPPMS) is a method for physical vapor deposition of thin films which is based on magnetron sputter deposition. HIPIMS utilises extremely high power densities of the order of kW⋅cm−2 in short pulses (impulses) of tens of microseconds at low duty cycle (on/off time ratio) of < 10%. Distinguishing features of HIPIMS are a high degree of ionisation of the sputtered metal and a high rate of molecular gas dissociation which result in high density of deposited films. The ionization and dissociation degree increase according to the peak cathode power. The limit is determined by the transition of the discharge from glow to arc phase. The peak power and the duty cycle are selected so as to maintain an average cathode power similar to conventional sputtering (1–10 W⋅cm−2).
HIPIMS is used for:
adhesion enhancing pretreatment of the substrate prior to coating deposition (substrate etching)
deposition of thin films with high microstructure density
Apply magnetic field to the target. Apply negative voltage.
Some electrons in the chamber bounce around.
Some electrons get trapped in the magnetic field of the target.
Add some cheap argon atoms. Electrons might collide with them to produce argon ions. Positive argon ions get attracted to the negative target.
There is a cascade effect and the target atoms/ions land on the substrate.
https://www.youtube.com/embed/L6ZIkmIVm6c?rel=0
Plasma glow, depends on the voltage, pressure, magnetic field, material.
Changing the shape/structure of what is grown. Changing the hardness.
Different plasma glows.
Pulsed laser deposition
https://andor.oxinst.com/learning/view/article/pulsed-laser-deposition
The technique of PLD has been used to deposit high quality films of materials for more than a decade. The technique uses high power laser pulses (typically ~108 Wcm-2) to melt, evaporate and ionize material from the surface of a target. This “ablation” event produces a transient, highly luminous plasma plume that expands rapidly away from the target surface. The ablated material is collected on an appropriately placed substrate upon which it condenses and the thin film grows. Applications of the technique range from the production of superconducting and insulating circuit components to improved wear and biocompatibility for medical applications. In spite of this widespread usage, the fundamental processes occurring during the transfer of material from target to substrate are not fully understood and are consequently the focus of much research.
In principle PLD is an extremely simple technique, which uses pulses of laser energy to remove material from the surface of a target, as shown schematically below.
https://www.youtube.com/watch?v=9u3-3v-i1P4
Different processes for different properties
Uses of superconductors: Low-loss power cables; Squids (superconducting quantum interference devices) – detect very low magnetic fields; quantum technology – electromagnets used in maglev trains, MRI (High voltage to low voltage, positive to negative), magnetic confinement fusion reactors (confinement of plasma consisting of positive ions and electrons), particle accelerators
Uses of superconductors – electromagnetism
Delocalised electrons move through the superconductor,
Why do the above diagrams imply the negatively charged electron is moving to the negative side?
They aren’t – the assumption goes back to Faraday’s idea about conventional current.
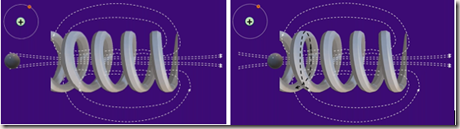
Make the copper wire into a coil (a solenoid)
Magnetic fields join together producing a field pattern that looks like the field pattern of a bar magnet with a central compact area
Drop a hydrogen ion (a proton) into a particle accelerator and it will be accelerated through the coil
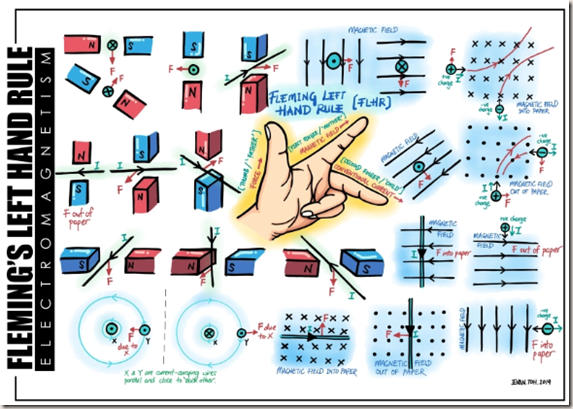
Fleming’s left-hand rule
https://evantoh23.wordpress.com/2019/05/01/flemings-left-hand-rule/
Fleming’s left-hand rule is used to work out the direction of the force on the charged particle. Align your second finger with the beam of particles (the current) remembering that it points the way positive particles flow, the opposite way to electron flow.
https://en.wikipedia.org/wiki/Magnetic_resonance_imaging
Magnetic resonance imaging (MRI) is a medical imaging technique used in radiology to form pictures of the anatomy and the physiological processes of the body. MRI scanners use strong magnetic fields, magnetic field gradients, and radio waves to generate images of the organs in the body. MRI does not involve X-rays or the use of ionizing radiation, which distinguishes it from CT and PET scans. MRI is a medical application of nuclear magnetic resonance (NMR). NMR can also be used for imaging in other NMR applications, such as NMR spectroscopy.
https://www.nhs.uk/conditions/mri-scan/
To perform a study, the person is positioned within an MRI scanner that forms a strong magnetic field around the area to be imaged. First, energy from an oscillating magnetic field is temporarily applied to the patient at the appropriate resonance frequency. Scanning with X and Y gradient coils cause a selected region of the patient to experience the exact magnetic field required for the energy to be absorbed. The excited atoms emit a radio frequency (RF) signal, which is measured by a receiving coil. The RF signal may be processed to deduce position information by looking at the changes in RF level and phase caused by varying the local magnetic field using gradient coils. As these coils are rapidly switched during the excitation and response to perform a moving line scan, they create the characteristic repetitive noise of an MRI scan as the windings move slightly due to magnetostriction. The contrast between different tissues is determined by the rate at which excited atoms return to the equilibrium state. Exogenous contrast agents may be given to the person to make the image clearer.
The major components of an MRI scanner are the main magnet, which polarizes the sample, the shim coils for correcting shifts in the homogeneity of the main magnetic field, the gradient system which is used to localize the region to be scanned and the RF system, which excites the sample and detects the resulting NMR signal. The whole system is controlled by one or more computers.
The human body is made up of a lot of hydrogen atoms (the human body is mainly water, which consists of two hydrogen atoms). A hydrogen ion is a proton with a particular spin and a magnetic moment parallel to it.
Normally these protons are randomly orientated so overall there is zero magnetic moment (they have been cancelled out)
Put a person in the MRI coil and the magnetic moments line up with the field.
An RF field changes some orientations. Remove the field leaving energy.
When the energy is released the moments line up the field again.
Fat releases energy faster than water. The time of release tells us whether the image is bone, fat, water etc.
With superconductors in the system you can remove the power supply. Very high magnetic fields are used.
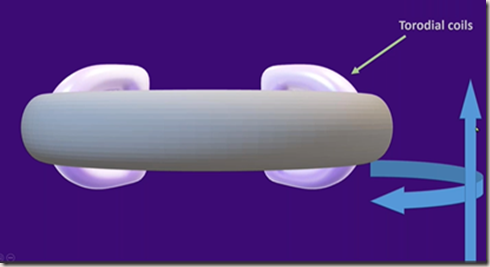
Uses of superconductors – particle accelerators
Bunches of coils formed into a doughnut shape
Lots of coils are used rather than one big one reduces the effect of Eddy currents.
Eddy currents are loops of electrical current induced within conductors by a changing magnetic field in the conductor according to Faraday’s law of induction. Eddy currents flow in closed loops within conductors, in planes perpendicular to the magnetic field.
By Lenz’s law, an eddy current creates a magnetic field that opposes the change in the magnetic field that created it, and thus eddy currents react back on the source of the magnetic field.
Charged particles are accelerated. The paths are defined and can’t the particles can’t escape.
Apply an external charge field to accelerate the particle. It’s relativistic mass increases.
Two coils side by side can give two beams of charged particles. They are accelerated in opposite directions and then allowed to collide
Other magnets are used to produce the change in direction that allows the collisions to occur.
Uses of superconductors – fusion energy
Two positive charges are brought close enough together they will fuse
The new particle formed is heavier and energy is released
https://en.wikipedia.org/wiki/Tokamak
A tokamak is a device which uses a powerful magnetic field to confine a hot plasma in the shape of a torus. The tokamak is one of several types of magnetic confinement devices being developed to produce controlled thermonuclear fusion power. As of 2016, it is the leading candidate for a practical fusion reactor
The tokamak reactor controls the nuclear fusion reaction
Inside the tokamak there is a magnetic field. ITER has 6
https://en.wikipedia.org/wiki/ITER
ITER (originally the International Thermonuclear Experimental Reactor) is an international nuclear fusion research and engineering megaproject, which will be the world’s largest magnetic confinement plasma physics experiment. It is an experimental tokamak nuclear fusion reactor that is being built next to the Cadarache facility in Saint-Paul-lès-Durance, in Provence, southern France.
There is a central coil.
Shaking material – particles collide by changing magnetic fields.
If the plasma is allowed to touch the wall, it would melt.
Fusion works but it might not be viable because of the cost and the fact that you get less energy out than you put in
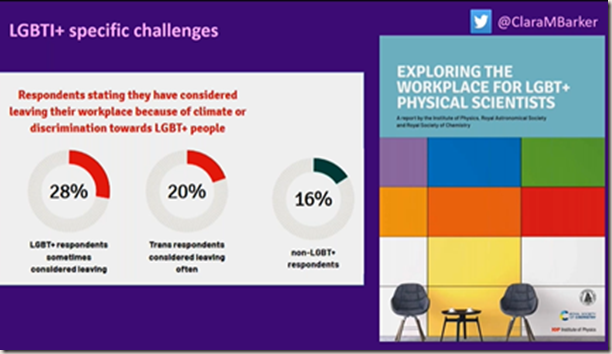
Diversity in STEMM
As a transgender woman diversity in STEMM is something that Dr Barker is passionate about
Since writing this article Dr Barker has read it. She was very complimentary and added some comments:
“We use both magnetron sputtering and pulsed laser deposition to make our thin films. I love that you included HiPIMS – which I have not talked about for a while but i spent 10 years researching. I am really hoping that I can get a grant to get a HiPIMS gemnerator in my new lab :)”
“I will also say, I know I said a few things wrong! So feel free to correct. it is one of those things that afterwards you wonder ‘why did I say that’, lol. I do have a series on the individual topics dicsussed, where I go into further detail on each area (though again, I know I am sure I made little mistakes – especially when talking about flux pinning!)”
Science with Clara https://www.youtube.com/playlist?list=PLgBDSWFBbq_uZAW02mUEJ9qu4W7n-14C8

























































One thought on “Scattering atoms, electrons and perceptions”